Production of liquefied carbon dioxide
Production of alcohol has long transformed into a modern technological industry, taking into account contemporary quality and standard requirements. One of the challenges faced by alcohol producers is the efficient management of carbon dioxide, which is produced during the fermentation process. The production of liquefied carbon dioxide is a crucial aspect of alcohol manufacturing as it enables energy storage, reduces environmental impact, and optimizes alcohol production.
Carbon dioxide (CO2), alongside ethyl alcohol, is a major product of alcohol fermentation.
Stoichiometric equation for alcohol fermentation:
C6H12O6 => 2C2H5OH + 2CO2↑ + 188 kJ
180.1 92.1 88
According to the stoichiometric equation provided above, for every 1 unit of ethyl alcohol produced, 0.9565 units of carbon dioxide are formed.
During fermentation, carbon dioxide bubbles passing through the mash layer become saturated with alcohol vapors and its volatile impurities. The fermentation gases consist of carbon dioxide, alcohol vapors, air, water vapor, alcohols, aldehydes, organic acids, and complex esters. Losses of alcohol due to evaporation depend on the alcohol strength and fermentation temperature. At a fermentation temperature of 30 °C, the average alcohol losses amount to 0.74% of the alcohol content in the mash.
The fermentation gases enter alcohol catchers where they are adsorbed by water and returned to the matured mash in the form of a water-alcohol mixture with a concentration of 1.5-7% vol. The fermentation gases, now free from ethyl alcohol and its impurities, are directed to the carbon dioxide production plant or released into the atmosphere.
To produce liquefied carbon dioxide from the fermentation products, it is sufficient to purify the gas from liquid droplets and organic impurities and prevent air from entering the gas stream. The purified and dried carbon dioxide is then sent to the cooling section of the plant for further liquefaction. Unfortunately, the quality of the obtained carbon dioxide from this raw material does not always meet consumer expectations.
Modern technology employs a two-stage purification process for carbon dioxide. In the first stage, it undergoes adsorption purification with activated charcoal in columns installed after the initial compression stage. In the second stage, it undergoes adsorption purification and drying first in a silica gel adsorber and then in a zeolite adsorber for deeper drying.
The technology of producing liquefied carbon dioxide is continuously evolving, aiming to improve its quality and meet the demands of consumers.
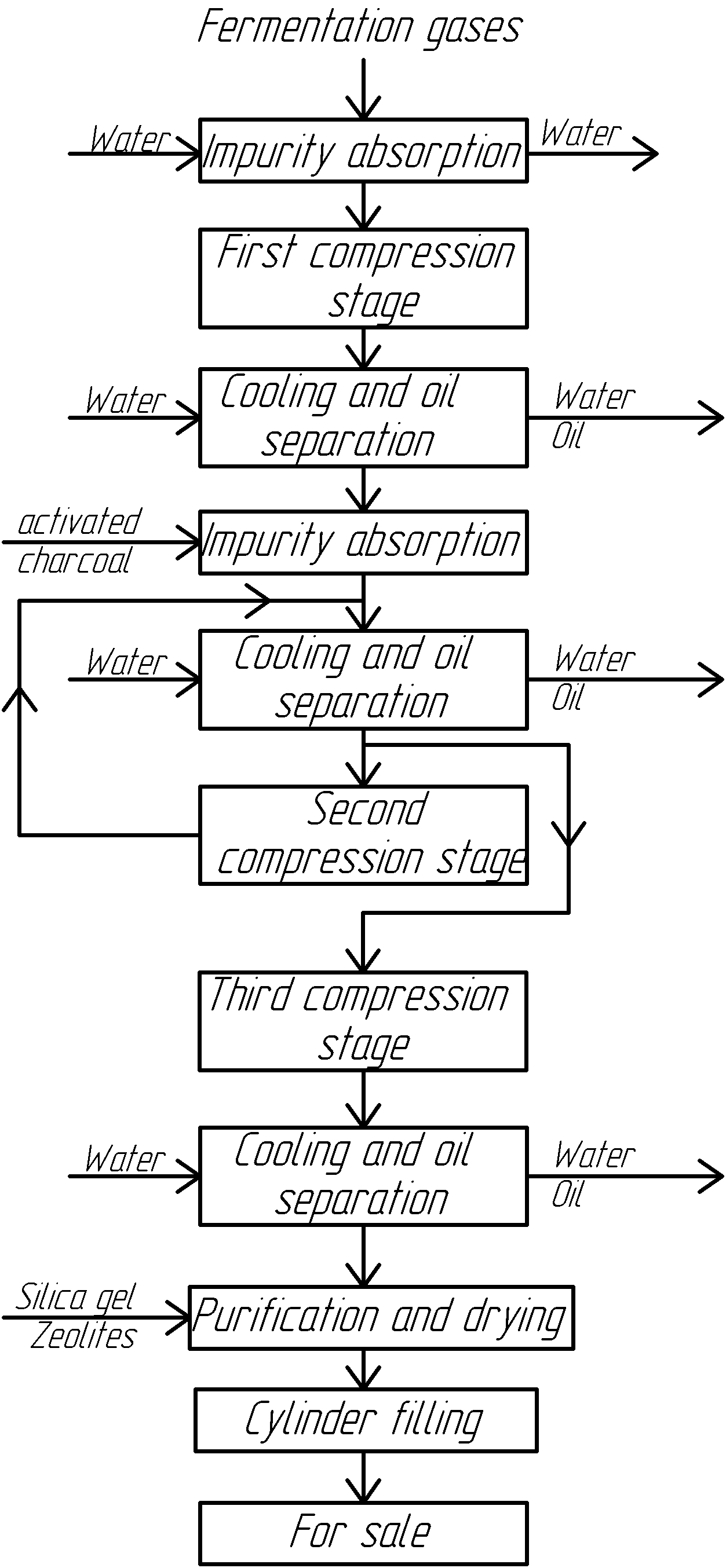
At alcohol plants, liquid carbon dioxide is obtained through a liquefaction process that involves using cold temperatures. The fundamental and improved technological scheme for producing liquid carbon dioxide is shown in the diagram above.
From the fermentation vessels, the fermentation gases are directed to a foam catcher and then to an alcohol catcher, where the gas is washed with water to remove organic impurities and cooled down. The cleaned gas is then sent to a water ring compressor, where it is further purified and cooled before being compressed to a pressure of 0.5 MPa in the first stage of a three-stage compressor and then entering a refrigeration unit. Oil separators are installed for the purification and drying of carbon dioxide before and after the refrigeration unit.
Afterward, the gas is purified using activated charcoal in two adsorbers, with one in operation while the other undergoes regeneration. Regeneration is done with heated carbon dioxide produced during throttling.
From the adsorbers, carbon dioxide enters the second stage of the compressor, where it is compressed to a pressure of 2.4…2.5 MPa, and then passes through the refrigeration unit and an oil separator before entering the third stage of the compressor. The gas, compressed to approximately 7 MPa, undergoes further purification and drying through a series of adsorbers filled with silica gel and zeolite.
In the condenser, the gas, after the third stage, condenses while releasing heat. The liquefied carbon dioxide is then transferred through a high-pressure receiver into steel cylinders placed on scales.
With this scheme, it is also possible to produce and store overcooled liquefied carbon dioxide without using cylinders. For this purpose, the liquid carbon dioxide is throttled from 6.5…7.0 to 0.8…1.2 MPa, resulting in an emulsion state. The liquid and gaseous phases are separated in a vortex distributor, with the gaseous phase making up about 47%. The liquid carbon dioxide flows through circular channels of the vortex chamber to a separation vessel and then to a storage vessel, isothermal storage, or a transport isothermal reservoir. The gaseous phase, on the other hand, flows through central openings of the vortex chamber and then through the corresponding communication channels to a mixer, where it combines with the gas supplied by the first stage of the compressor. The gas then proceeds to the second stage of the compressor.
The amount of liquid carbon dioxide entering the isothermal storage is monitored by a level indicator, and pressure is measured by a pressure gauge. The maximum filling of the isothermal storage is 85…90% of the geometric volume. The parameters of the liquid carbon dioxide in the isothermal reservoir are as follows: pressure of 0.8…1.2 MPa, temperature ranging from -43.5 to -33.3 °C, heat of vaporization of 326…309 kJ/kg, and density of 1130.8…1087.8 kg/m³.
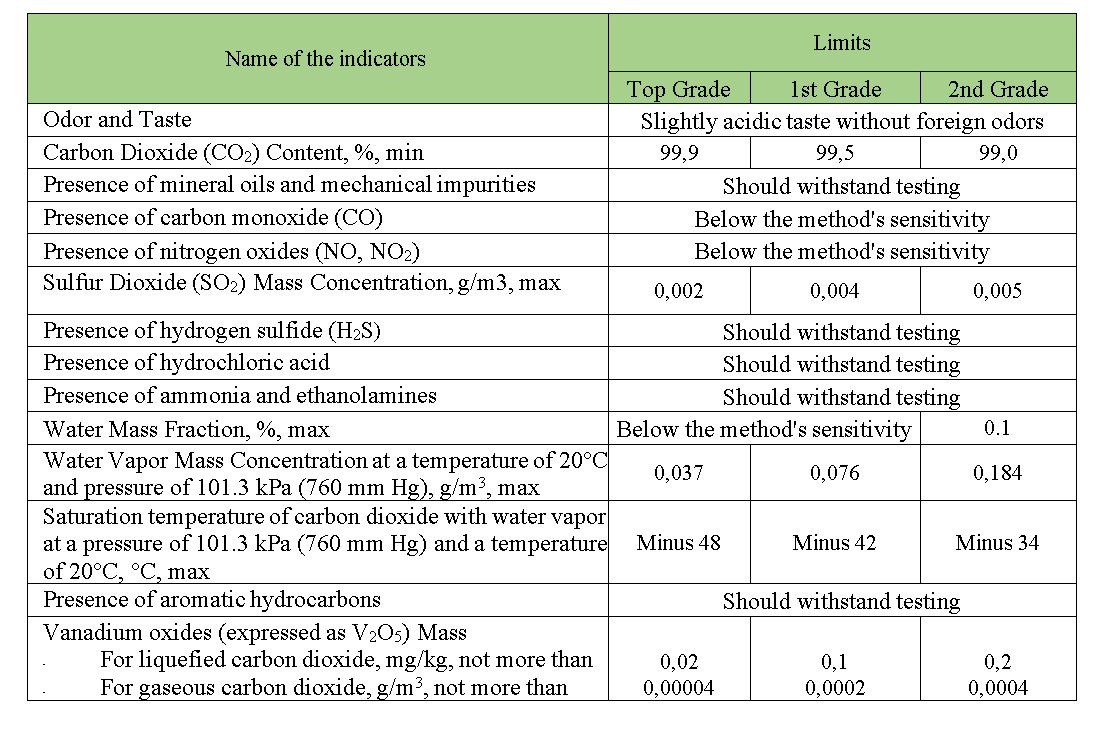
Liquid carbon dioxide can be utilized in the production of dry ice. The quality of liquid carbon dioxide obtained from alcohol fermentation gases is regulated by the DSTU 4817:2007 standard. Organoleptic and physicochemical characteristics for food-grade and non-food-grade carbon dioxide are provided in the table below.