Evaporation of molasses stillage
At most alcohol distilleries that process molasses, the distillation residue is discharged onto fields for filtration. If the quantity of residue is significant (12 m3 per 100 liters of produced denatured alcohol), the utilization of large areas of fertile land for cultivating agricultural crops becomes impractical. Additionally, the surrounding air becomes polluted with decomposition products of organic compounds.
One of the existing methods for molasses residue utilization is its evaporation. The chemical composition of the evaporated post-alcohol residue is presented in Table 1.
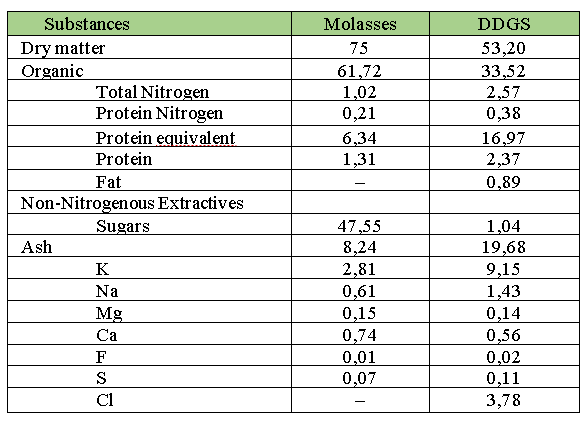
Table 1 – Comparative Chemical Composition of Molasses and Evaporated Post-Alcohol Residue
Organic acids, glycerol, and other compounds present in small quantities in the evaporated molasses residue have little practical significance.
The color substances of the residue, which possess surface-active properties, make it suitable for use as a diluent and plasticizer in cement and concrete production.
Nitrogen-containing substances, colloids, and potassium salts determine the suitability of evaporated post-yeast residue as a fertilizer, especially in combination with superphosphate.
Evaporated residue can be transported over considerable distances – up to 2,000 km. Various schemes for evaporation stations and evaporator designs have been developed for molasses residue evaporation.
The residue is evaporated in 3…4-stage evaporators with steam reuse. The residue passes through several stages or chambers in sequence. Live steam is supplied only to the first stage. Subsequent stages are heated by secondary steam from the previous stage. The temperature in each subsequent stage is reduced by decreasing the pressure at which the residue boils.
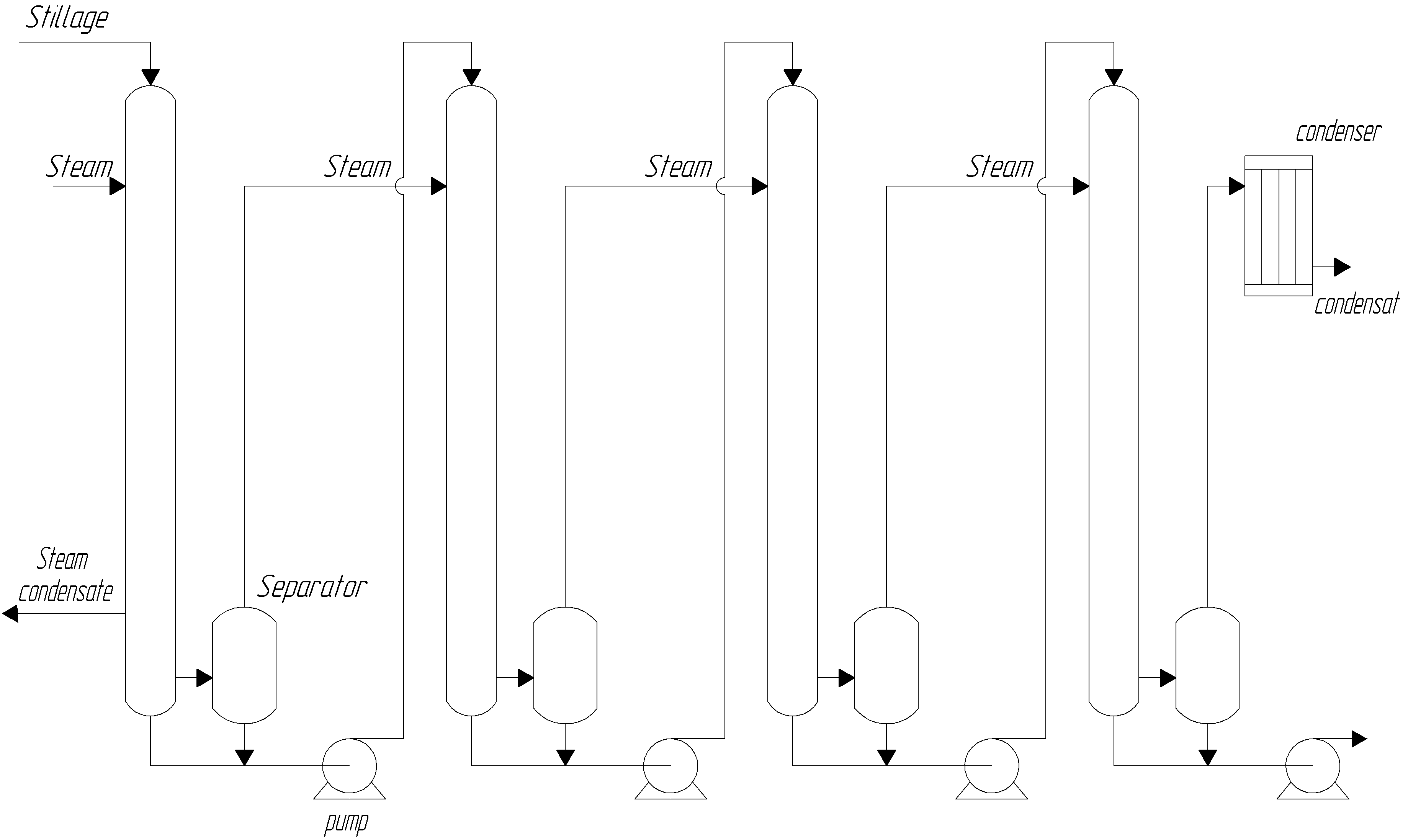
The secondary steam from the last chamber is directed into a barometric condenser, while the non-condensing gases are evacuated by a vacuum pump, creating a pressure reduction down to 84 kPa.
The maximum temperature during the evaporation of the residue should not exceed 140… 142°C; otherwise, the breakdown (caramelization) of sugars and amino acids occurs. Vitamins also degrade at high temperatures. These processes lead to a decrease in the biological value of the evaporated residue

